*All other brand or product names located in this site are Trademarks of their respective holders.
Montelukast Sodium Tablets
NDC Number :
60505-3562-03
Material Number :
59547
Active Ingredient :
Montelukast Sodium
Bioequivalent to * :
SINGULAIR®
Therapeutic Class (AHFS) :
Leukotriene Modifiers
Strength :
10MG
Pack Size (Form) :
30 TABLET (BOTTLE)
Dosage Form :
Tablet, Film Coated
Scored :
NO
Color :
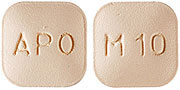
BEIGE
Shape :
ROUNDED, SQUARE-SHAPED, BICONVEX FILM-COATED TABLETS
Markings :
ENGRAVED WITH "APO" ON ONE SIDE AND "M10" ON THE OTHER SIDE
Route of Administration :
Oral
Storage Condition :
Controlled Room Temperature (68 - 77 degrees F)
Minimum Order Quantity :
12
Multiple Order Quantity :
12
Bar Code to Unit-Of-Use :
Yes
Product Resources :
Prescribing Information (344 KB)

not to scale
Packaging Information :
| Length | Width | Height | Weight | Units/Pack | |
|---|---|---|---|---|---|
| Unit | 1.8 in | 1.8 in | 3.3 in | 0.07 lb | N/A |
| Inner | 7.1 in | 5.3 in | 3.3 in | 0.9 lb | 12 |
| Case | 11.7 in | 8.1 in | 9.3 in | 5.9 lb | 72 |
 United
States |
United
States | 


